|
Apimostinel
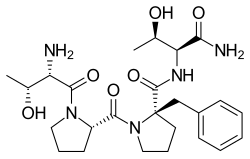
Apimostinel (GATE-202, formerly NRX-1074) is an investigational antidepressant, acting as a novel and selective modulator of the NMDA receptor.[1][2][3][4] It is currently under development for the acute treatment of major depressive disorder (MDD) by Gate Neurosciences, and previously by Naurex and Allergan.[5][6][7] As of February 2015, an intravenous formulation of apimostinel has completed a phase IIa clinical trial for MDD.[5][8] Similar to rapastinel (GLYX-13), its mechanism of action acts through a unique binding site on the NMDA receptor, independent of the glycine site, to modulate receptor activity and enhance NMDAR-mediated synaptic plasticity.[9] However, apimostinel is 1000-fold more potent in vitro and is intended as an improved, follow-up drug to rapastinel.[2][5] Similar to rapastinel, apimostinel is an amidated tetrapeptide, but has been structurally modified, via the addition of a benzyl group, to enhance its metabolic stability and pharmacokinetic profile. The drug has shown rapid and potent antidepressant effects in pre-clinical models of depression.[5] In addition, similarly to rapastinel, it is well tolerated and lacks the schizophrenia-like psychotomimetic effects of NMDA receptor antagonists such as ketamine.[5] See alsoReferences
External links |
||||||||||||||||||||||||||||||||||||||||||||