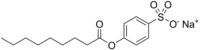
Sodium nonanoyloxybenzenesulfonate
Names
Preferred IUPAC name
Sodium 4-(nonanoyloxy)benzene-1-sulfonate
Other names
4-Sulfophenyl nonanoate sodium salt; Sodium p -nonanoyloxybenzenesulfonate; p -(Nonanoyloxy)benzenesulfonic acid sodium salt; p -Sodiosulfophenyl nonanoate
Identifiers
Abbreviations
NOBS
ChemSpider
UNII
InChI=1S/C15H22O5S.Na/c1-2-3-4-5-6-7-12-15(16)20-13-10-8-9-11-14(13)21(17,18)19;/h8-11H,2-7,12H2,1H3,(H,17,18,19);/q;+1/p-1
Y Key: QSKQNALVHFTOQX-UHFFFAOYSA-M
Y InChI=1/C15H22O5S.Na/c1-2-3-4-5-6-7-12-15(16)20-13-10-8-9-11-14(13)21(17,18)19;/h8-11H,2-7,12H2,1H3,(H,17,18,19);/q;+1/p-1
Key: QSKQNALVHFTOQX-REWHXWOFAO
[Na+].O=C(Oc1ccccc1S([O-])(=O)=O)CCCCCCCC
Properties
C 15 H 21 Na O 5 S
Molar mass
−1
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
Chemical compound
Sodium nonanoyloxybenzenesulfonate (NOBS ) is an important component of laundry detergents and bleaches . It is known as a bleach activator for active oxygen sources, allowing formulas containing hydrogen peroxide releasing chemicals (specifically sodium perborate , sodium percarbonate , sodium perphosphate , sodium persulfate , and urea peroxide ) to effect bleaching at lower temperatures.[ 1]
Synthesis
NOBS is formed by the reaction of nonanoic acid (or its esters ) with phenol followed by aromatic sulfonation using SO3 to form a sulfonic acid at the para-position.
Bleach activation
NOBS was developed by Procter & Gamble in 1983[ 2] [ 3] [ 4] TAED , which is the predominant bleach activator used in Europe, NOBS is efficient at much lower temperatures. At 20 °C NOBS is 100 times more soluble than TAED in water.[ 5] peroxynonanoic acid (a peroxy acid ) and releases the leaving group sodium 4-hydroxybenzene sulfonate, which is an inert by-product.
References
^ Kuzel, P.; Lieser, T. (1990). "Bleach systems". Tenside, Surfactants, Detergents . 27 (1): 23– 8. doi :10.1515/tsd-1990-270109 . S2CID 235325050 . ^ Chung, S. Y.; Spadini, G. L. (1983). US4412934 . ^ Arno Cahn (30 January 1994). Proceedings of the 3rd World Conference on Detergents: Global Perspectives 64– 70. ISBN 978-0-935315-52-3 ^ Hirschen, M. (2005). Handbook of Detergents Part C: Analysis . Marcel Dekker. pp. 439– 470. ISBN 9780824703516 ^ Reinhardt, G.; Borchers, G. (2009). Handbook of Detergents, Part E: Applications . CRC Press. ISBN 9781574447576